
If we assume further that the initial momentum has not changed until the position measurement, we can speak of a definite momentum until the time of the position measurement. So the three measurements are known precisely. 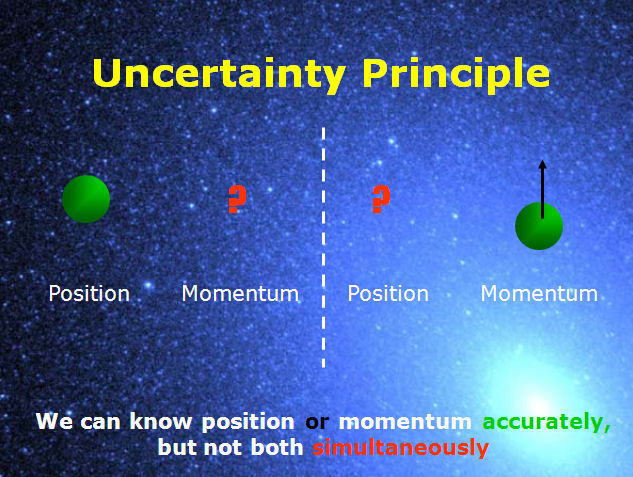
And finally the final momentum (pf) is measured. We assume the initial momentum of the electron (pi) is known precisely, it has been measured in a previous experiment. 
According to Heisenberg’s ‘measurement=meaning principle’, one must also specify, in the given context, what the meaning of the phrase ‘momentum of the electron’ is, in order to make sense of the claim that this momentum is changed by the position measurement. In its present form it is an epistemological principle, since it limits what we can know about the electron.īut this argument is incomplete. This is the first formulation of the uncertainty principle. Thus, at the moment when the position of the particle is accurately known, its momentum cannot be precisely determined. In such a collision, the electron suffers a recoil which disturbs its momentum. The interaction of the electron and the illuminating light should then be considered as a collision of at least one photon with the electron. However, with smaller wavelength light such as gamma rays, the Compton Effect becomes too significant to be ignored. This enables us to be able to view the electron and know its exact position. Given that the energy contained in a photon of light is inversely proportional to the wavelength of light, it means that for more accurate measurement of the position of the electron, very high energy photons are used. From classical mechanics we do understand that to view an object or particle for that matter, there must be an interaction between the light and the particle. To make very precise measurements one needs to use light of lower wavelength such as gamma rays. The accuracy of such a measurement rests on the wavelength of light that is used. The best example that Heisenberg offered was the measurement of the position of the electron by the use of a microscope. We will call this assumption the ‘measurement=meaning principle.’ The Uncertainty principle was born of a disagreement between Heisenberg and Schrödinger, each of them defending the particle and the wave views of quantum mechanics (how this led to the uncertainty principle is of no importance in the current work.) Heisenberg adopted an operational assumption: terms like ‘the position of a particle’ have meaning only if one specifies a suitable experiment by which ‘the position of a particle’ can be measured. Heisenberg observed for example that it was impossible to measure both momentum and position of a particle within the same experiment. In quantum mechanics, this is not always true. During a single experiment to measure volume, we can measure values for height, length and width. For example in the computation of speed of an object, we must simultaneously have the value for distance and time in the same experiment. As such, I will do away with all the mathematical derivations of the principle I will post those in a later post.Ĭlassical Mechanics, that is Newtonian Physics, presupposes that exact simultaneous values can be assigned to all physical quantities. It has therefore dawned on me to simplify it for digestion by the general public and physicists that are just getting to know what it is all about. The problem stems from the inability to understand this principle. The people who ask this question know exactly the definition of this principle. Over the past not so few weeks I have been asked what the uncertainty principle is.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
